|
RT-PCR of undifferentiated ES cell markers. This graph showed the average of three independent experiments. The number of colonies was counted and compared with the results of SNL feeder. The graph shows the percentage of TRA-1-60 positive colonies. 201B7 iPS cells at passage number 19 were plated on each feeder, and incubated for 6 days. Images of iPS cells maintained on each fibroblast at passage number 10 and 19. HDF can maintain self-renewal of established human iPS cells.Ī. In addition, reverse transcription polymerase chain reaction (RT-PCR) showed that the expression of ES cell marker genes such as OCT3/4, SOX2 and NANOG were equally to those of H9 ES cells at passage 19 ( Fig. In iPS cells at passage 2 after switching onto various HDF feeders, no significant re-activation of transgenes was observed ( Fig. 
No significant differences were observed in the plating efficiencies ( Fig. The percentage of TRA-1-60 (a marker for undifferentiated ES cells and iPS cells) positive colonies was similar among different human fibroblasts and SNL cells ( Fig. All the five cell lines of feeder cells were supportive for undifferentiated growth of iPS cells at least 19 additional passages ( Fig. The passage number of iPS cells was 20 at this point. Then, we plated 201B7 iPS cell line derived from 1388 fibroblasts onto these feeder cells with standard density (1∶5 dilutions). To examine whether human fibroblasts support self-renewal of human iPS cells, we treated four independent human fibroblast lines (1388, 1392, 1503 and NHDF see Table S1) and SNL cells with mitomycin C, and seeded them on culture plates ( Fig. Since iPS cells are generated from fibroblasts, it would be ideal if the same fibroblasts can be used for the generation and maintenance of iPS cells. However, one still have to concern about existence of unidentified pathogens, such as viruses and prions in these non-autologous feeders. 
To avoid these issues, human fibroblasts from neonatal foreskin or ES cell-derived fibroblast-like were used to support self-renewal of human ES cells –. Although feeder-free culture of human ES cells has been reported, it may lead to chromosomal instabilities of human ES cells. In fact, non-human sialic acid N-glycolylneuraminic acid (Neu5Gc), which is potentially immunogenic, was detected on the surface of human ES cells maintained on MEF feeder. However, usage of mouse feeder cells may transfer exogenous antigens, unknown viruses, or zoonotic pathogens to iPS cells. Human pluripotent stem cells, both embryonic stem (ES) cells and induced Pluripotent Stem (iPS) cells, are generally maintained on mouse embryonic fibroblasts (MEF), which are mitotically inactivated by treatment with mitomycin C or γ-ray irradiation –. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT), Japan Science and Technology Agency (JST), National Institute of Biomedical Innovation (NIBIO), Japan Society for the Promotion of Science (JSPS). Received: AugAccepted: OctoPublished: December 2, 2009Ĭopyright: © 2009 Takahashi et al. PLoS ONE 4(12):Įditor: Martin Pera, University of Southern California, United States of America 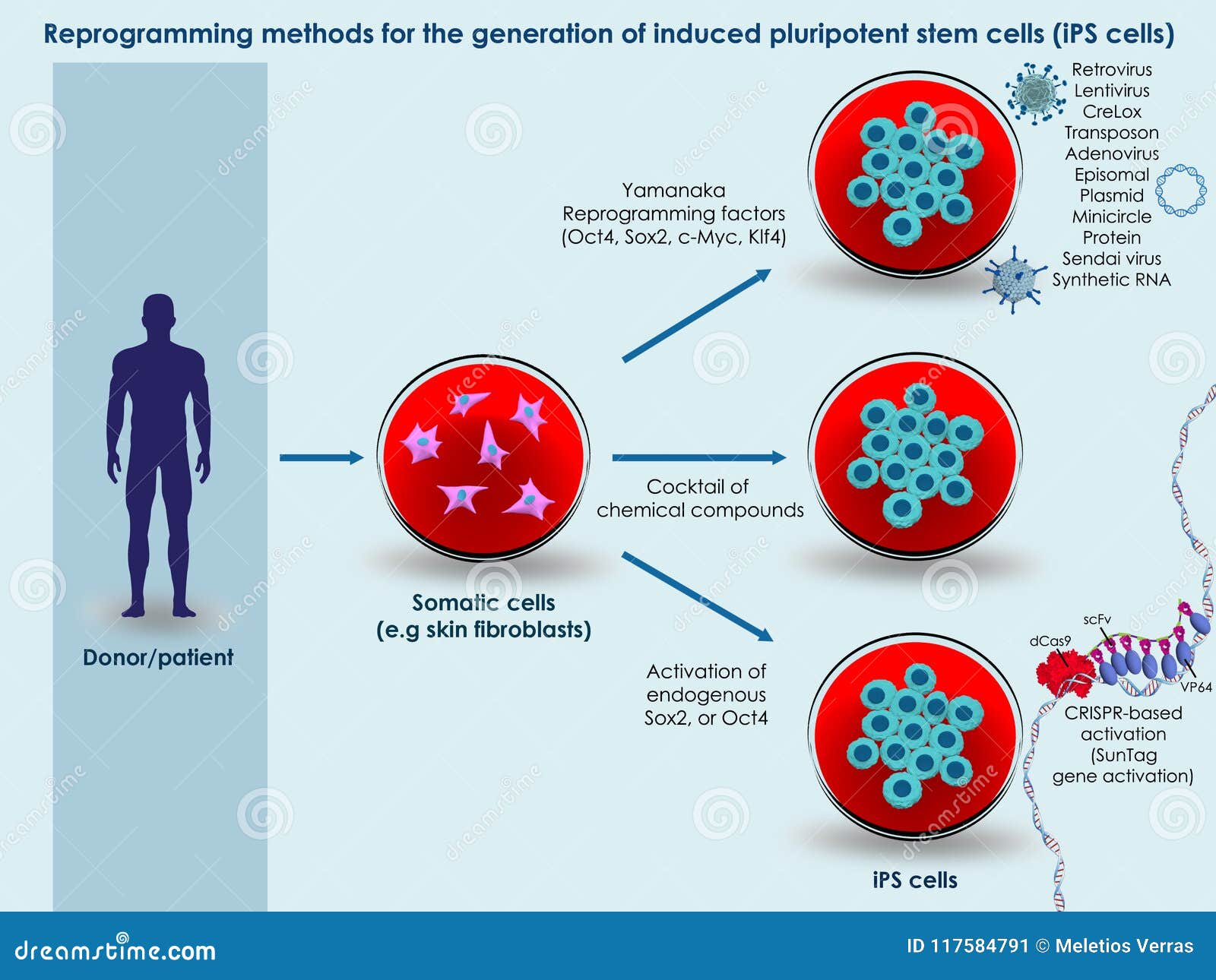
Citation: Takahashi K, Narita M, Yokura M, Ichisaka T, Yamanaka S (2009) Human Induced Pluripotent Stem Cells on Autologous Feeders.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
